Welcome to Ethide Laboratories
We’ll Ensure You Always Get The Best Result
Our Company’s mission is to deliver a personalized approach to contract testing services so our customers can focus on their vision of bringing their products to market faster.

Feel secure with your contract testing services.
Ethide Labs brings a wealth of experience having over 60 years in the medical device and allied industries.
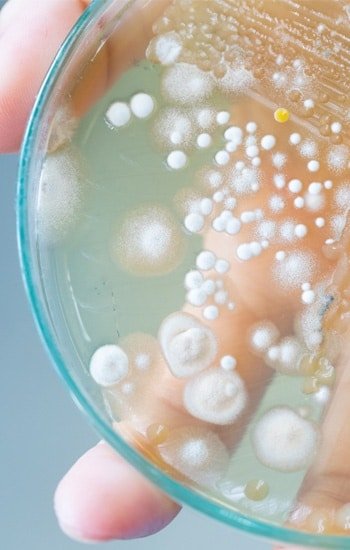
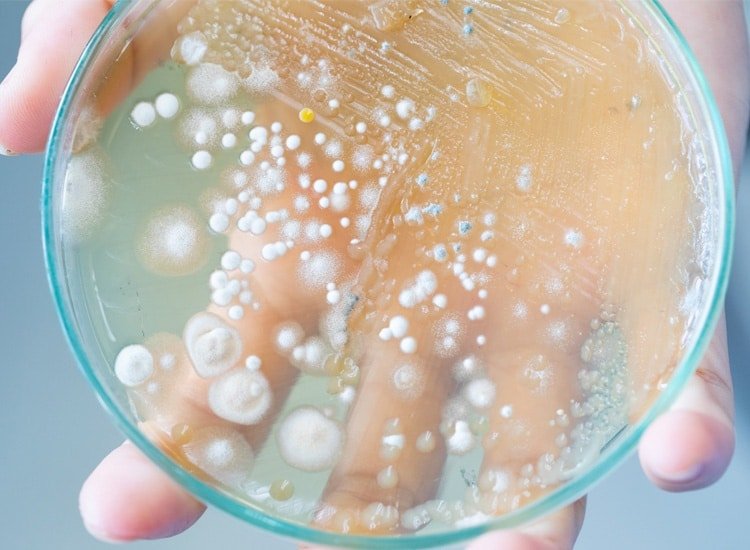
Population of viable microorganisms on or in product and/or sterile barrier system.
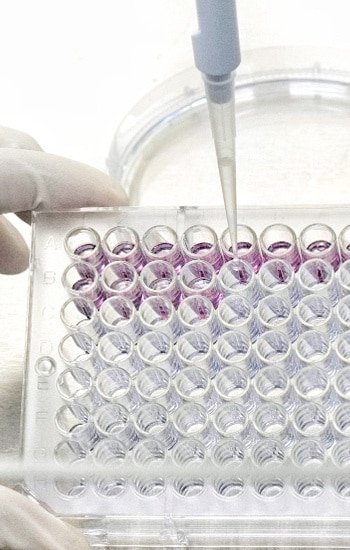
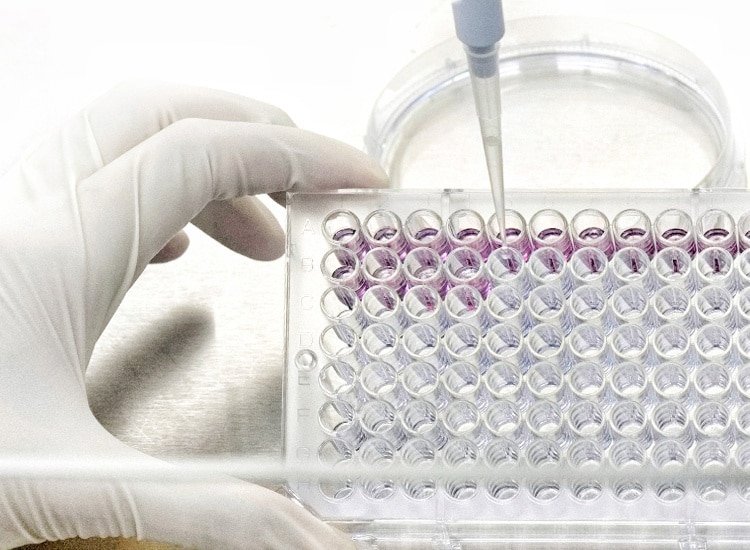
Validation or requalification to determine the presence or absence of microorganisms on product.
Population and identification of microorganisms present on a product or device.
Amounts of Ethylene Oxide residuals present on product post sterilization.
Population of viable microorganisms on or in product and/or sterile barrier system.
Validation or requalification to determine the presence or absence of microorganisms on product.
Population and identification of microorganisms present on a product or device.
Amounts of Ethylene Oxide residuals present on product post sterilization.
Determination of seal strength and integrity of seals used in packaging.
Evaluation of the toxicity of medical devices, products, and materials.
Evaluation of products for the presence of endotoxins, like gram-negative bacteria.
Determination of seal strength and integrity of seals used in packaging.
Evaluation of the toxicity of medical devices, products, and other materials.
Evaluation of products for the presence of endotoxins, like gram-negative bacteria.
Ethide Labs is a contract testing organization specializing in Ethylene Oxide (EO) residual and microbiology testing. We also offer bioburden testing, sterility testing, package integrity testing & environmental monitoring services for medical device companies.
Ethide Labs is an ISO 13485 certified facility. All our laboratory equipment is on a routine, documented maintenance and calibration program. Rigorous internal audits for key processes are also conducted regularly by qualified personnel.
Get a Quote
Are you looking for expert guidance, better communication and faster testing turnaround times compared to your current contract testing partners? Or are you simply looking for relevant testing strategies? You have come to the right place. We can be your guide and help you navigate complex regulatory pathway for your device.

We Employ The Latest Technical Guidance & Expertise
At Ethide Laboratories, our staff participates in technical committees to keep up on latest FDA guidance and reference standards in the industry.
“The choices we make, make a difference. Our service principles improve our customers’ lives, protect our environment and have an impact on individuals who work to protect human health. We invest in our capabilities, our processes, our people and our customers to both sustain our business and reach our goals. As we strive for excellence, we measure and trend our performance, not only to satisfy our customer requirements but – more importantly – to identify continuous improvement opportunities.”

Sarath Koruprolu,
Ethide Labs CEO
Why Choose Us
We provide our contract testing services with a personal approach and exell in service quality, accuracy & efficiency.
High Quality Lab
Ethide Labs follows a rigorous program to be ISO 13485:2016 certified testing laboratory registered for microbiological, biological and EO residual testing for the medical device industry.
Unmatched Expertise
Ethide brings a wealth of experience having over 50 years in the medical device and allied industries.
Precise Results
All testing procedures have been developed referencing current national and international standards to ensure valid test data.
Qualified Staff
All technical staff are degreed scientists and active members of the American Society for Microbiology.
Information you need to know about contract testing services: EO residual, bioburden and microbiology testing, sterilization validation, accelerated aging, and more!
How Comprehensive Product Testing Expertise Reduces Risk for Medtech Manufacturers
When the lifesaving and lifechanging products you make are going into patients’ bodies, there is no room for error. Every medical device manufacturer knows this…
Accelerating Time to Market By Streamlining Medical Device Testing
One of the biggest challenges in medical device manufacturing is simply getting the product to market quickly. Speed to launch isn’t just about competitive advantage;…
The Benefits of Outsourcing Medical Device Product Testing
As a medical device manufacturer, you know that every product you bring to market must meet rigorous testing standards to ensure patient safety.
Why People Trust Us
Know why our customers choose us as their contract testing services provider.

Christopher Lavallee
Clean Room Certs, Inc.
“Ethide personnel have been instrumental in using their direct knowledge of USP-797 to help educate our customers and ensure they receive the latest guidance and inputs that could affect their businesses. Ethide has helped our customers best determine the types of testing that would most suite them based on the direct nature of those customers work related functions. CCS would highly recommend Ethide Laboratories to other medical device manufacturing companies for their continued ability to quickly fill our sometimes-short notices requests for sampling supplies.”
Get One Step Ahead Of Competition
Are you looking for expert guidance, better communication and faster testing turnaround times compared to your current contract testing partners? Or are you simply looking for relevant testing strategies? You have come to the right place. We can be your guide and help you navigate complex regulatory pathway for your device.